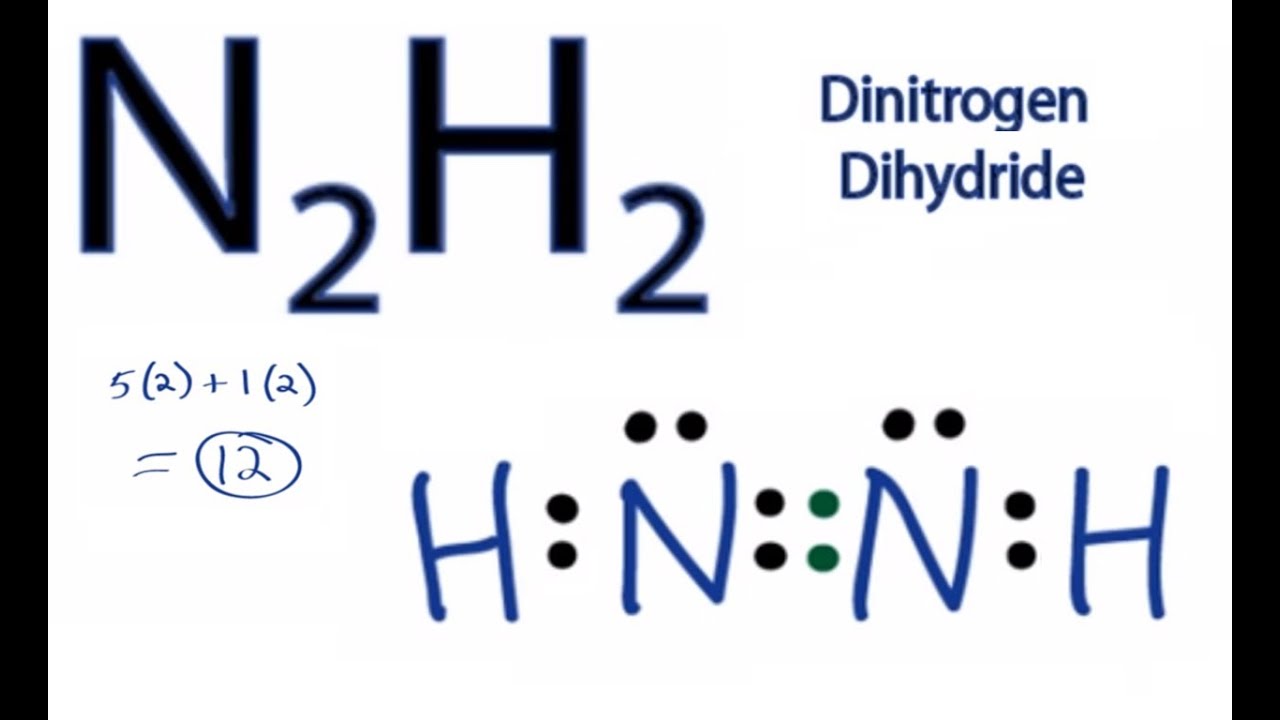N2H2 is polar molecule with london dispersion forces,dipole dipole forces and H bonding .True or false?How?

N2H2 Molecular Geometry, Bond Angles & Electron Geometry (Diimide) | N2H2 Molecular Geometry, Bond Angles & Electron Geometry (Diimide) Molecules like N2H2 can be a little tricky because although they might seem

Part A Predict whether each of the following molecules is polar or nonpolar Drag the appropriate items to - brainly.com